- Home
- Migraine Treatments
- Nerivio Migraine Treatment
The Nerivio Migraine Treatment Device: Q & A With CEO Alon Ironi
 Nerivio Migraine Treatment Device is FDA-Cleared @migrainesavvy
Nerivio Migraine Treatment Device is FDA-Cleared @migrainesavvy
I got to ask Alon Ironi, the CEO and founder of Theranica (the parent company of Nerivio) some questions about the new wireless device that’s helping migraine sufferers all over the world.
It’s been named one of the best inventions of 2019... the smartphone-controlled device uses electrical stimulation to block pain signals from reaching the brain. And in 2 studies, pain relief was found to be comparable to taking a triptan! WOW!
Here's what you need to know before we get to the Q&A with Nerivio's founder and CEO.
Contents:
How It Works | FDA-Cleared | For Acute Treatment | Who Can Use It | Side Effects | Q & A with CEO | Benefits | Cost | Key Takeaways | Where To Find It
The Nerivio Migraine Treatment Device Is...
|
|
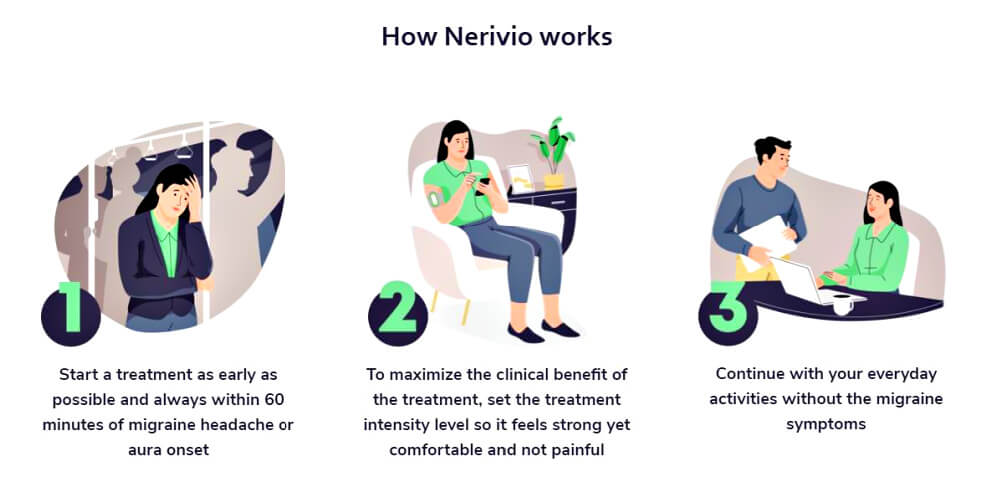
*The device is placed on the upper arm which is a great feature for people with migraine who don't want to electronically stimulate an already painful head and neck. Plus you can wear it discretely and no one with notice that you are enduring an attack. [1]
And what is most impressive is that 2 studies proved the relief from this device were at a "comparable level to the relief of triptans." [1]
66.7% of patients achieve pain relief
37.4% of patients achieve pain freedom
~ David Yarnitsky, et al. (Headache, 2019)
Nerivio Is "FDA-Cleared"
In case you want to know about this distinction between FDA approved and FDA-cleared or authorized...
FDA approval is only given to Class III medical devices. In contrast, Nerivio went through a 510k submission and received clearance/approval to market from the FDA.
The FDA market authorization was based on the results of a prospective, randomized, double-blind, placebo-controlled, multi-center pivotal study, where 252 patients from 12 clinics used the non-invasive wearable to treat their migraine attacks.
Theranica received FDA De Novo authorization to market Nerivio in the United States in May of 2019, as a first-in-category product.
Who can use the nerivio migraine treatment device?
I think these points are important to mention:
 The Nerivio App and Armband
The Nerivio App and ArmbandNerivio Is Used for Adults Episodic Migraine: Nerivio is used for adults with episodic migraine with or without aura, as shown in this study. [3]
Heart Problems: people with migraine, with a history of heart problems are good candidates here because the less-expensive triptan medications just aren’t safe for them to take. [1]
Children: sadly, kids have fewer treatment options than adults. "The company website reports Theranica is vowed to have its risk-free, effective Nerivio cleared for adolescents and kids, and provide them with a comfortable, affordable, easy-to-use, rapid solution for acute Migraine. It’s just not cleared yet." [1]
What are the side effects?
Is this going to hurt? That's your first question... right.
Most people didn’t report any side effects, and the reported effects disappeared within 24 hours. The FDA considers Nerivio a “minimal risk” treatment, meaning they don’t expect to see any serious side effects from its use.
~ Dr. Sara Crystal, MD, Neurologist, Cove Medical Director [2]
Here are some of the side effects of the Nerivio migraine treatment device that have been reported:
- feeling of warmth
- itching or tingling
- mild pain in the arm, shoulders, or neck
- muscle spasm
- temporary numbness in the arm or hand
And now to the Q&A...
Migraine Devices Vs. Traditional Treatments
Q&A with Alon Ironi, CEO and Co-Founder of Theranica
Besides medication side-effects, what are the benefits of migraine devices versus traditional treatments?
Although migraine is the third most common disease worldwide, affecting 14.7 percent of the global population, many are still searching for effective long-term pain relief. With the rapid pace of the 21st Century, people living with migraine are not only in need of affordable solutions with little-to-no side effects, but treatments that can easily be used on the go, for extended periods of time.
Typically, migraine is treated with over the counter pain relievers (OTCs) or medications specifically prescribed for migraine. However, neither of these options are ideal long-term solutions. OTCs are less effective than prescription medication, often leading to over consumption and resulting in medication overuse headaches. Prescription medications can cause harmful side effects, and the high price tag of many drugs serves as a deterrent.
Migraine devices are offering an alternative solution to the standard migraine treatment, with technology that brings affordable, low side-effect and drug free therapies to combat pain and related symptoms of a migraine attack. What is most notable is some migraine devices, including Nerivio, have proven to be as effective and safe as traditional medications, opening the door for a new future of migraine treatment. Not all devices are the same of course. Different devices have unique qualities of evidence-based clinical data, and the comfort level varies on the body. It is best to consult with the physician about the anticipated efficacy of each device.
Certain migraine devices also bring a unique added value to the therapeutic space: data. With life in the 21st Century revolving around technology, migraine devices that connect to smartphone applications, such as Nerivio, can help active users keep track of and better manage their migraine episodes by adding an additional layer of information. Smartphone applications include migraine diaries and harness the power of AI to develop personal analytics that can easily be shared with care teams to inform treatment plans and improve treatment outcomes. Additionally, unlike migraine medications that are often administered with broad dosages, users of therapeutic devices can control the strength of each migraine treatment to find one that suits best with their tolerance levels.
Ultimately, with the goal of providing relief to those living with migraine, drug-free devices are quickly growing in popularity. Not only can devices serve as a first-line treatment, replacing drugs, they can also be used alongside medications.
Today, there are a variety of migraine devices such as GammaCore, sTMS, eNeura. What is different about Nerivio?
Most migraine devices today are placed on the head or neck, which can be cumbersome. Some are less effective than prescription drugs, and they come at varying prices.
From day one, Theranica set out to develop a product that was as effective as leading prescription medications, with a high safety profile, at an affordable price. Nerivio was developed to provide people with an affordable long-term solution. Offered at $99, the Nerivio only needs to be replaced after 12 uses.
Nerivio is uniquely placed on the upper arm and is the first smartphone-controlled migraine wearable to receive De Novo clearance from the U.S. Food and Drug Administration.
Nerivio uses Remote Electrical Neuromodulation, utilizing an endogenous neurological mechanism called Conditioned Pain Modulation, which has been extensively studied and proven to be as effective as pharmacological options. Moreover, the data show that the efficacy of Nerivio is sustained over a 24 to 48-hour period, which is quite rare with a non-pharmacological therapy.
What is perhaps even more important, is that Nerivio shows consistency across multiple treatments, offering a safe, long-term therapeutic alternative with minimal side effects and no contraindications.
Since our market launch earlier this year, Nerivio has been used by almost 7,000 patients across the United States, who have administered almost 50,000 treatments, and this number is rapidly growing. Our device is currently approved for acute migraine treatment for adults and is currently under FDA review for expansion to chronic migraine. In the meantime, we are also working on gathering data for further expansion to extend clearance to adolescents.
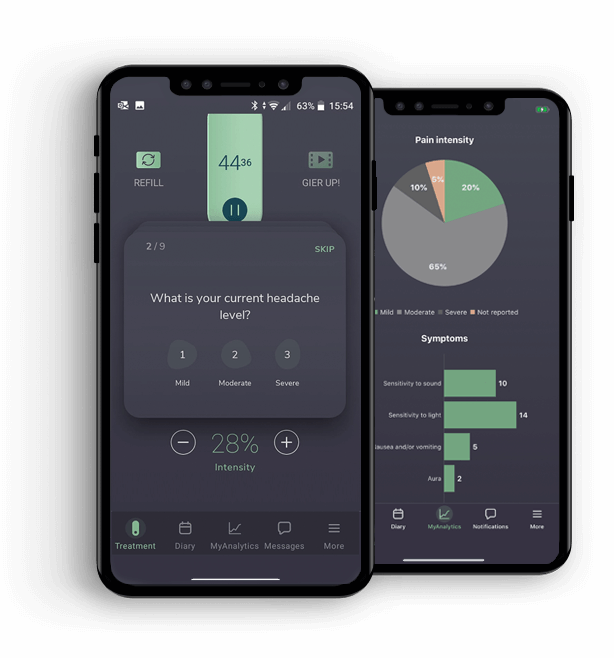 The Nerivio app also has a diary. An essential tool to help you prevent attacks @migrainesavvy
The Nerivio app also has a diary. An essential tool to help you prevent attacks @migrainesavvyKey Takeaways
- This technology brings affordable, low side-effect and drug free therapies to combat pain and related symptoms of a migraine attack.
- Provides us with data - smartphone applications include migraine diaries and harness the power of AI to develop personal analytics that can easily be shared with care teams to inform treatment plans and improve treatment outcomes.
- The Nerivio migraine treatment device can be used alongside almost any medication.
- Offers an affordable price $99 but needs to be replaced after 12 uses.
- The efficacy of the Nerivio migraine treatment device is sustained over a 24 to 48-hour period.
Some of the other devices on the market today, also approved by the FDA include: Cefaly, electroCore’s GammaCore, and eNeura sTMS.
Where You Can Find It
Here's where you can find the Nerivio Migraine Treatment Device:
Nerivio is available on two telemedicine platforms: Cove and UpScript. These platforms allow you to get relief to your door using the online platform.
** Theranica Announces Nerivio will be Available in Select European Countries Starting in 2021 **
Nerivio® Smartphone-Controlled Wearable for acute treatment of migraine has been granted CE Mark for use in Europe.
YAY!
BE INFORMED. STAY INFORMED. JOIN THE MAILING LIST.
Ready to take the next step?
Choose the next step that fits where you are right now.
MIGRAINE TREATMENTS Related Articles
Nerivio Migraine Treatment References (and great articles):
1. Dumas, P. (2020) New Wireless Armband for Migraines Now Available – How to Get It. Available [online]: https://www.migraineagain.com/armband-for-migraines/
2. Cove.com (2020) Everything to Know About Nerivio, the New Innovative Migraine Treatment. Available [online]: https://www.withcove.com/learn/migraine-treatment-device-nerivio
3. Yarnitsky, D., Volokh, L., et al. Neurology (March 2017) Nonpainful remote electrical stimulation alleviates episodic migraine pain. Available [online] at: https://n.neurology.org/content/88/13/1250.short
Nerivio Migraine Treatment Images: Theranica